MtoZ Biolabs has launched the protein purity and homogeneity characterization service which utilizes multi-platform technology to accurately analyze the purity of intact proteins, the consistency of subunit composition, and the homogeneity of samples. This service is broadly applied in protein-based research and development, structural studies, and batch-to-batch protein quality monitoring, providing a reliable data foundation for molecular mass assessment and experimental progression.
Overview
Protein purity and homogeneity describe the proportion of target intact protein within a sample and the consistency of its molecular species, indicating the presence or absence of contaminant proteins, mis-folded variants, aggregates, or degradation products. These attributes directly determine identification accuracy, structural integrity confidence, and the comparability of results across batches, making them essential early indicators in protein characterization. This assay plays a key role in antibody engineering, recombinant protein development, structural biology, and biotherapeutic protein quality assessment, serving as a critical basis for confirming protein physicochemical properties and steady-state folding behavior.
Protein Purity and Homogeneity Characterization Service at MtoZ Biolabs
MtoZ Biolabs, utilizing multi-platform integrated technologies, provides customers with analytical testing centered on protein purity and sample homogeneity.
-
Protein purity quantification
-
Assessment of contaminant protein residual levels
-
Determination of heterogeneity interference caused by protein aggregates or degradation products
-
Evaluation of purity consistency across different batches
-
Monitoring of molecular species heterogeneity in samples
Our analysis delivers a comprehensive understanding of protein sample purity and spectral uniformity, offering researchers a reliable data basis for advancing functional protein studies.
Technical Platforms for Protein Purity and Homogeneity Characterization Service
1. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
Used to evaluate whether the protein band is singular, supporting assessment of sample purity.
2. Size Exclusion Chromatography (SEC)
Used to assess protein solution homogeneity and aggregation level by resolving monomer and aggregate peaks.
3. Gel Permeation Chromatography (GPC)
Used to confirm whether protein components exhibit a single, uniform distribution based on molecular volume separation.
4. High Performance Liquid Chromatography (HPLC)
Used to quantify protein purity by measuring the primary peak area ratio while resolving secondary or impurity components.
5. Liquid Chromatography-Mass Spectrometry (LC-MS)
Used to obtain high-accuracy protein mass and composition patterns, supporting confirmation of target protein purity and batch uniformity.
6. Circular Dichroism (CD) Spectroscopy
Used to monitor global folding envelopes, supporting auxiliary evaluation of
7. Nuclear Magnetic Resonance (NMR) Spectroscopy
Used to evaluate protein structural uniformity through resonance line shapes and chemical shift dispersion patterns.
8. Dynamic Light Scattering (DLS)
Used to evaluate solution homogeneity and detect protein aggregation through hydrodynamic size distribution profiles.
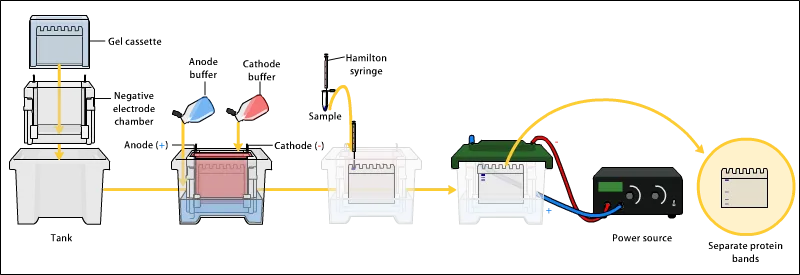
Figure 1. The Analysis Workflow of Protein Purity by SDS-PAGE.
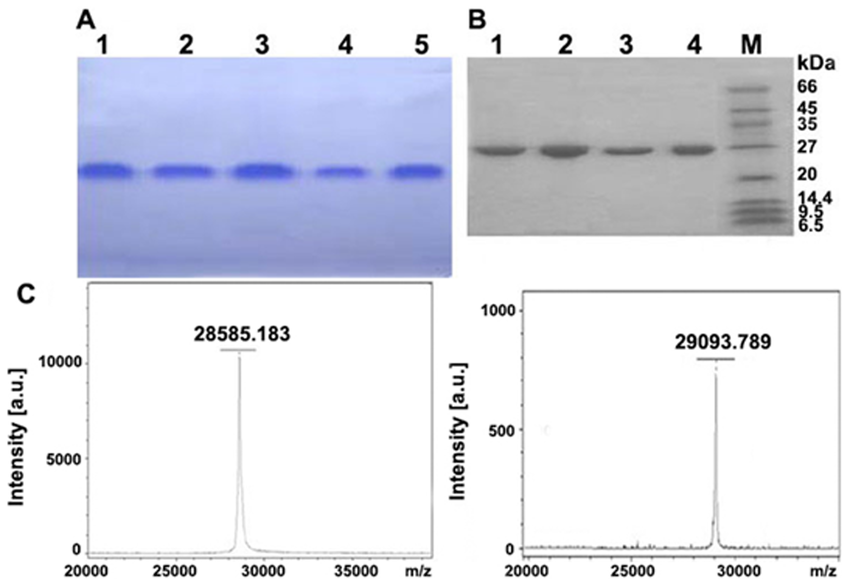
Meng, Y. et al. PLOS One, 2014.
Figure 2. Size, Homogeneity and Subunit Structure Analysis of Two Purified Proteins.
Why Choose MtoZ Biolabs?
1. Reliable Separation
Mature multi-platform workflows ensure stable isolation of the primary protein from interfering components.
2. High-Confidence Purity Readouts
Standardized calibration systems deliver low-deviation, cross-batch comparable purity quantification.
3. Robust Homogeneity Profiling
Validated, well-established processes provide strong consistency across samples and production batches.
4. Low Sample Consumption
Efficient detection supports low-input protein characterization and micro-scale systems.
5. One-Stop Delivery
Includes sample evaluation, method integration, data processing, and formal report generation.
Applications of Protein Purity and Homogeneity Characterization Service
1. Protein Engineering Screening
Used to evaluate whether peak profiles of engineered allosteric variants or fusion proteins converge and whether sample purity meets specifications.
2. Formulation Assessment
Used to determine the effects of buffer salts, stabilizers, or excipient ratio changes on protein solution uniformity, supporting steady-state judgments during formulation screening.
3. Biomarker Candidate Screening
Used to assess whether candidate proteins in biomarker discovery or mechanistic studies exhibit a high-purity, low-interference dominant peak distribution.
4. Protein Integrity Evaluation
Used to determine whether truncation, misfolding, aggregation, or degradation products substantially impact sample purity or homogeneity.
5. Purification Process Optimization
Used to evaluate the potential for purity improvement and interference reduction in purified proteins from spleen, tissues, or biofluids, enabling iterative refinement of purification strategies.
Deliverables
- Comprehensive Experimental Details
- Materials, Instruments, and Methods
- Protein Purity and Homogeneity Results Table
- Key Spectral or Curve Data
- Bioinformatics Analysis
- Raw Data Files
FAQ
Q1: What types of samples are suitable?
A1: Suitable samples include soluble, clear, and high-purity protein preparations such as recombinant proteins, antibodies, fusion proteins, and affinity- or SEC-purified complexes. Samples should avoid substantial particulates, non-volatile salts, and strongly absorbing matrix components that may interfere with spectral or chromatographic readouts.
Q2: What is the service general workflow?
A2:
Q3: What data formats are provided?
A3: MtoZ Biolabs provides multiple standardized deliverables, including
-
Quantitative results tables for purity and homogeneity (XLSX/CSV)
-
Electrophoresis and chromatography images (PNG/TIFF)
-
Raw MS files in instrument-compatible formats
-
Key curves or spectral datasets from NMR, CD, or DLS analyses (TXT/CSV or image files)
-
A comprehensive characterization report (PDF)
If special analytical requirements exist, data formats can be customized according to project specifications.
Q4: How should I prepare my samples?
A4: To ensure optimal identification results, it is recommended to prepare samples from the following aspects:
- Purity: Solutions must be free of visible precipitation or turbidity, with controlled levels of salt, glycerol, and surfactants
- Storage: Maintain at 4℃ for short-term or at -80℃ for long-term, avoiding repeated freeze-thaw cycles
- Transport: Ship under cold-chain conditions (ice packs or dry ice) using leak-proof, sealed packaging to minimize particle introduction
- Additional Information: Include protein source, buffer composition, and concentration estimates
For more information, please refer to Sample Submission Guidelines for Proteomics, Sample Submission Guidelines for Metabolomics.
Start Your Project with MtoZ Biolabs
Contact us to discuss your experimental design or request a quote. Whether you are investigating protein folding quality, conformational uniformity, batch-to-batch consistency, or process-induced purity shifts, MtoZ Biolabs can deliver precise analytical support.
